Expediting Discovery and Development
How did Mindbreeze help a pharmaceutical company speed up time to market by 50%?
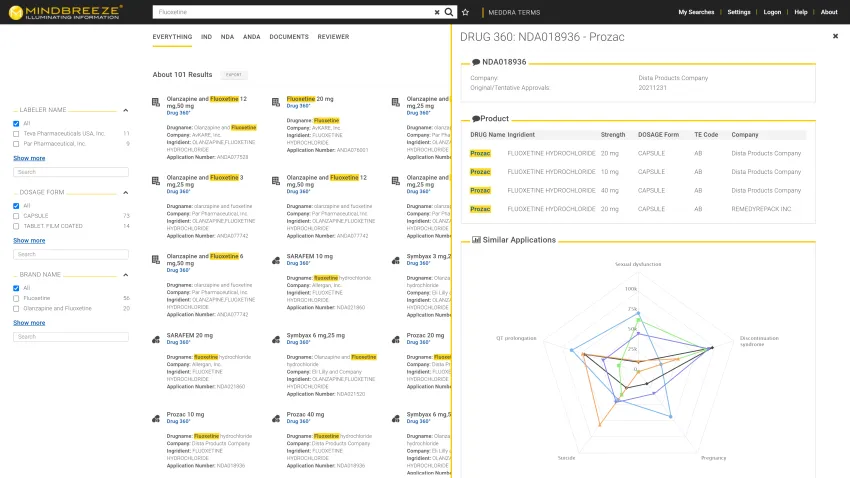
Mindbreeze's intelligent document processing and knowledge extraction capabilities streamlined processes for a major pharmaceutical organization – fast research, more knowledge, and informed decision-making.
Problem
Life science organizations often face challenges managing and utilizing their data effectively to make quicker and more informed decisions. Access to scattered data across various systems makes it practically impossible for researchers and authorities to make life-changing discoveries in a highly regulated industry.
This organization had:
- 6,500 different product categories
- More than 17,000 employees (all needing critical information)
- Millions of indexed documents
The organization needed to analyze clinical trial data, drug information, and review applications faster and more consistently.
Solution
Mindbreeze indexed existing internal data (from Documentum, such as action letters, review recommendations, and information requests) and external data (such as eCTD backbone: forms, trial data) and linked it with structured data from the Oracle database.
AI Features Used:
- Search-driven analytics for autosuggestions based on user search history, popular queries, and document keywords
- Entity Recognition Service for automatic extraction of necessary information from large documents and datasets.
- Semantic Relations Service to identify and extract correlations of existing information and provide relevant results on a priority basis.
Data Sources:
- Internal data from Documentum, an enterprise content management system
- External data from trials and medical forms
- Oracle Database